Can AI Replace Animal Testing?
Animal studies are still the default. Animal studies have long been the backbone of drug and chemical safety testing. They give researchers rich, multi-layered data with parameters like blood chemistry, organ function, and physiological responses which help predict how a substance might behave in humans. But they come with problems. They’re expensive, time-consuming, ethically problematic, and not always predictive of human outcomes. With increasing pressure to reduce animal use (the “3Rs”: Replacement, Reduction, Refinement), researchers are actively searching for alternatives.
Enter AnimalGAN: a virtual animal model
This study introduces AnimalGAN, an artificial intelligence model built using a type of generative AI called a Generative Adversarial Network (GAN). Instead of just predicting whether a drug is “toxic” or “not toxic,” AnimalGAN does something more ambitious — it generates full synthetic datasets that mimic real animal experiments. Specifically, it can simulate: 38 clinical pathology measurements (like liver enzymes and blood markers), can do this across different doses as well as over multiple time points. In simplicity, it can create animals in which to experiment on.
How it works (without getting too technical)
AnimalGAN was trained on data from over 6,000 real rats exposed to 110 different compounds. It learns the relationship between: the chemical structure of a compound, the dose and duration of exposure as well as the resulting biological responses. Then, when given a new compound, it generates realistic biological outcomes as if the experiment had actually been run in reality.
Does it actually match real animal data?
When tested against real experimental data: The synthetic results closely matched actual measurements, similarity between real and generated data was extremely high, error rates were much lower than background variability. This occurred even when challenged with different drug classes than what it was originally tested on, newer drugs and chemically distinct compounds.
Better than traditional AI approaches
Most AI models in toxicology (like QSAR models) predict single outcomes , for example, whether a compound is toxic for a single parameter. AnimalGAN however is different in the case that it can predict entire biological profiles at once as well as capture the complexity of real physiology. When compared directly, AnimalGAN outperformed 12 standard QSAR models and produced more accurate predictions across all measured biomarkers. This essentially means that AI can move from simple classification of individual parameters to actually simulating biology in a virtual space.
Can it actually replace animal testing?
Not entirely yet, but it is getting close. It has helped in identifying liver toxicity and kidney toxicity where AnimalGAN agreed with studies over ~96% of the time. This level of similarity between virtual findings and real findings shows that animal testing could be reduced and could serve as a screening tool before in vivo studies. It would also help prioritize compounds for testing.
A major advantage: detecting rare toxic effects
One of the biggest limitations of both animal studies and clinical trials is sample size. This is especially the case with rare adverse events like Idiosyncratic drug induced liver injury (iDILI), and these events are rarely captured during testing because of their low prevalence during testing. AnimalGAN solves this by simulating massive sample sizes of over 100,000 rats. In this paper they compared the findings for 3 drugs that cause this rare liver injury and AnimalGAN was able to predict the prevalence accurately.
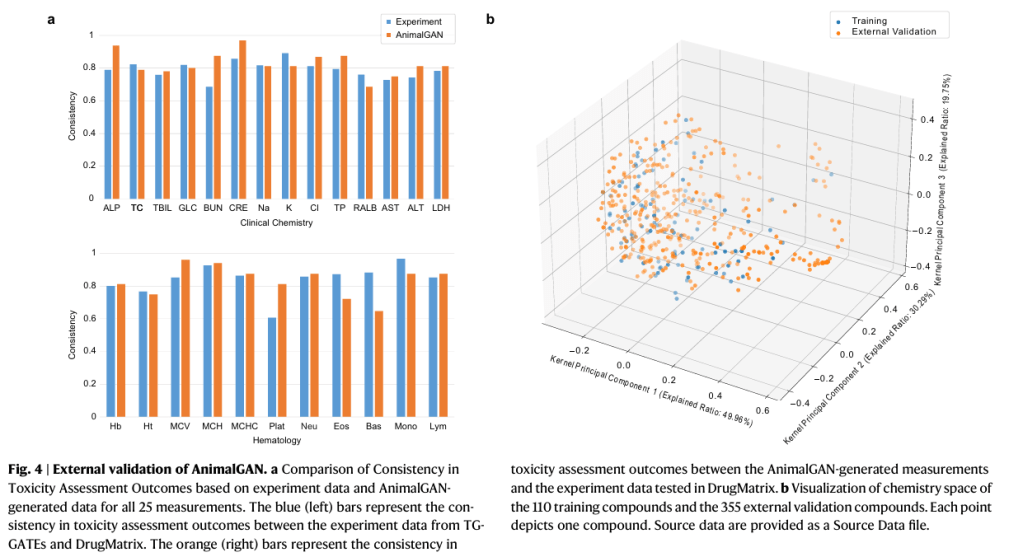
Real-world validation (and limitations)
When tested against an external dataset (DrugMatrix): AnimalGAN achieved 83% agreement with the real experiment results. This level of agreement is quite similar to comparing real world data sets among themselves, thus suggests that AnimalGAN may be as consistent as biological models. Some of the limitations are that: it was trained on a limited chemical space, performance outside that space is still uncertain, biological complexity is not fully captured.
Conclusion
There are thousands of new chemicals introduced every day. Testing all of them using traditional animal studies is simply not feasible, but more so, reducing the number of animals needed for testing would be a major shift in animal welfare and ethics. AnimalGAN could provide a rapid, high-throughput and ethically favourable alternative that can be run in tandem with real life testing.